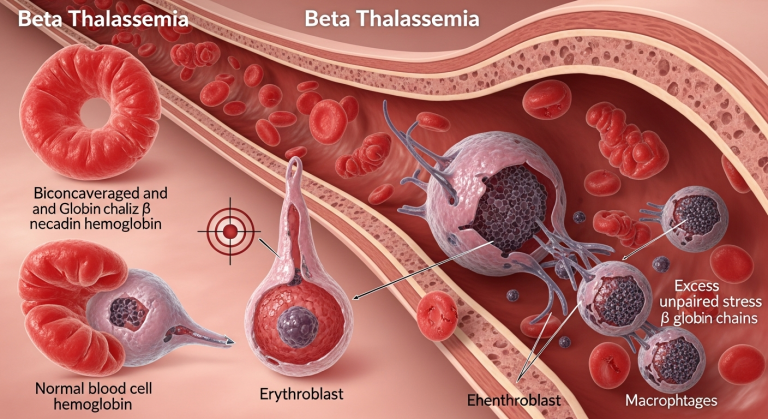
Beta thalassemia pathophysiology involves reduced or absent beta-globin chain production, causing an imbalance in hemoglobin synthesis. This leads to ineffective erythropoiesis, chronic anemia, and red blood cell destruction, ultimately resulting in complications like bone deformities and organ damage.
Molecular Pathophysiology
 Understanding beta thalassemia pathophysiology requires looking closely at the molecular level, where the imbalance of globin chains sets a destructive process in motion. These microscopic changes ultimately drive the clinical severity of the disease.
Understanding beta thalassemia pathophysiology requires looking closely at the molecular level, where the imbalance of globin chains sets a destructive process in motion. These microscopic changes ultimately drive the clinical severity of the disease.
Reduced or Absent Beta-Globin Synthesis
Because of the genetic mutations described above, the cells cannot produce enough beta-globin. This creates an immediate deficit in functional Hemoglobin A. Without sufficient hemoglobin, red blood cells lose their ability to efficiently carry oxygen to body tissues, leading to chronic anemia—one of the hallmark features of beta thalassemia pathophysiology.
Alpha-Globin Chain Excess
The lack of beta-globin leaves alpha-globin chains without binding partners. Since alpha-globin production continues at a normal rate, a significant imbalance develops. These excess alpha chains are highly unstable and cannot remain freely dissolved within the red blood cell.
Formation of Inclusion Bodies
Because the excess alpha-globin chains cannot form proper hemoglobin molecules, they begin to precipitate inside red blood cells. These aggregates form solid protein clumps known as inclusion bodies. They attach to the cell membrane, causing oxidative damage and disrupting the structural integrity of the cell. This process makes red blood cells rigid and fragile.
Membrane Damage and Premature Cell Destruction
The oxidative stress caused by inclusion bodies damages the red blood cell membrane, reducing its flexibility. As a result, these abnormal cells are either destroyed prematurely in the bone marrow (ineffective erythropoiesis) or removed by the spleen in circulation (hemolysis). This continuous destruction significantly worsens anemia and is a central mechanism in beta thalassemia pathophysiology.
Additional Key Point: Ineffective Erythropoiesis
A major feature of beta thalassemia pathophysiology is ineffective erythropoiesis. Although the bone marrow works overtime to produce more red blood cells in response to anemia, many developing cells die before reaching maturity due to globin imbalance and cellular damage. This not only fails to correct anemia but also leads to bone marrow expansion and skeletal abnormalities over time.
Cellular Pathophysiology: Erythropoiesis
Erythropoiesis is the process by which the bone marrow produces new red blood cells. In beta thalassemia pathophysiology, this process becomes highly inefficient and destructive, contributing significantly to disease severity.
Ineffective Erythropoiesis in the Bone Marrow
Normally, red blood cell precursors mature in the bone marrow before being released into the bloodstream. However, in beta thalassemia, the accumulation of excess alpha-globin inclusion bodies causes severe intracellular damage. The resulting oxidative stress triggers apoptosis (programmed cell death), destroying a large proportion of developing cells before they can mature.
Premature Destruction of Red Blood Cell Precursors
Because these precursor cells are eliminated so early, only a small number of red blood cells successfully enter circulation. This condition, known as ineffective erythropoiesis, is a defining feature of beta thalassemia pathophysiology and a major contributor to chronic anemia.
Bone Marrow Expansion and Deformities
In response to persistent anemia, the kidneys increase the production of erythropoietin, a hormone that stimulates red blood cell formation. This causes the bone marrow to expand in an attempt to compensate. Over time, this excessive expansion leads to thinning of the bone cortex and structural weakness. Patients—especially children with untreated thalassemia major—may develop characteristic skeletal deformities, including facial bone changes and increased fracture risk.
Extramedullary Hematopoiesis
When the bone marrow can no longer meet the body’s demand for red blood cells, the body activates alternative sites of blood production, such as the liver and spleen. This process is known as extramedullary hematopoiesis. It often leads to enlargement of these organs (hepatosplenomegaly), which can cause additional complications and discomfort.
Additional Key Point: Chronic Hypoxia and Systemic Effects
A critical consequence of beta thalassemia pathophysiology at the cellular level is chronic hypoxia (low oxygen supply to tissues). Due to the persistent shortage of functional red blood cells, vital organs do not receive adequate oxygen. Over time, this can impair growth, delay development in children, and contribute to long-term organ dysfunction if not properly managed.
Peripheral Blood Pathophysiology
 The red blood cells that manage to survive the bone marrow and enter the circulation still face significant challenges.
The red blood cells that manage to survive the bone marrow and enter the circulation still face significant challenges.
Hemolysis of Mature Red Blood Cells
The mature red blood cells that reach the bloodstream are inherently defective. Their cell membranes are damaged by the alpha-globin precipitates, making them rigid and fragile. As these defective cells navigate the narrow capillaries of the circulatory system, they easily rupture. This premature destruction of red blood cells is known as hemolysis.
Characteristics of the Anemia
The combination of ineffective erythropoiesis and peripheral hemolysis results in a profound anemia. Under a microscope, the red blood cells appear distinctly abnormal. They are microcytic (unusually small) and hypochromic (pale, due to the lack of hemoglobin).
The Spleen’s Role in Cell Destruction
The spleen acts as the body’s blood filter, tasked with removing old or damaged red blood cells. Because the red blood cells in beta thalassemia are misshapen and rigid, the spleen recognizes them as defective and aggressively destroys them. This rapid clearance further worsens the patient’s anemia.
Systemic Complications of Beta Thalassemia
The chronic lack of oxygen and the ongoing destruction of blood cells eventually impact nearly every organ system in the body.
Iron Overload (Hemosiderosis)
Iron overload is the most life-threatening complication of severe beta thalassemia. It occurs through two primary mechanisms:
- From Transfusions: Patients with severe thalassemia require frequent blood transfusions to survive. Each unit of transfused blood contains a significant amount of iron. Since the human body lacks a natural mechanism to excrete excess iron, it rapidly accumulates.
- From Increased Intestinal Absorption: The chronic state of anemia and ineffective erythropoiesis suppresses hepcidin, a hormone that regulates iron. This suppression tricks the gut into absorbing excessive amounts of dietary iron.
Over time, this excess iron deposits into vital organs—particularly the liver, heart, and endocrine glands—causing severe tissue damage and organ failure.
Splenomegaly and Hypersplenism
Working overtime to clear defective red blood cells causes the spleen to become massively enlarged, a condition known as splenomegaly. An oversized spleen can become overactive (hypersplenism), inappropriately destroying healthy white blood cells and platelets alongside the damaged red blood cells.
Bone Abnormalities and Osteoporosis
The continuous expansion of the bone marrow weakens the skeletal system. Patients frequently suffer from reduced bone mineral density, leading to osteopenia and osteoporosis, even at a young age.
Endocrine and Cardiac Complications
Iron deposition in the endocrine glands can lead to a host of hormonal issues, including insulin-dependent diabetes, hypothyroidism, and hypogonadism (which delays puberty and causes infertility). Furthermore, iron accumulation in the heart tissue can cause arrhythmias and heart failure, which historically has been the leading cause of mortality in patients with thalassemia major.
Increased Susceptibility to Infections
Patients face a higher risk of serious infections. This vulnerability stems from several factors, including the immunosuppressive effects of severe iron overload, the potential transmission of blood-borne pathogens through frequent transfusions, and the surgical removal of the spleen, which plays a vital role in immune defense.
Diagnosing Beta Thalassemia
 Early and accurate diagnosis is critical for managing the disease effectively. Medical professionals rely on a combination of laboratory tests to confirm the condition.
Early and accurate diagnosis is critical for managing the disease effectively. Medical professionals rely on a combination of laboratory tests to confirm the condition.
Complete Blood Count (CBC)
A CBC provides a snapshot of the patient’s blood profile. In beta thalassemia, the CBC typically reveals low hemoglobin levels, a reduced mean corpuscular volume (MCV), and low mean corpuscular hemoglobin (MCH).
Hemoglobin Electrophoresis and HPLC
To determine the specific types of hemoglobin present in the blood, doctors use techniques like hemoglobin electrophoresis or high-performance liquid chromatography (HPLC). These tests can confirm the diagnosis by showing elevated levels of Hemoglobin A2 and Hemoglobin F, alongside a significant decrease or total absence of normal Hemoglobin A.
Genetic Testing
DNA analysis provides the definitive diagnosis. By sequencing the HBB gene, geneticists can identify the exact mutations present. This is particularly valuable for family planning, prenatal diagnosis, and predicting the clinical severity of the disease.
Principles of Management
While beta thalassemia pathophysiology explains the underlying disease mechanisms, effective management focuses on controlling symptoms, preventing complications, and improving quality of life. Advances in modern medicine have transformed beta thalassemia from a fatal childhood disorder into a manageable chronic condition.
Blood Transfusions and Iron Chelation
The cornerstone of treatment for severe cases is a regular blood transfusion regimen, typically required every two to four weeks. These transfusions help maintain adequate hemoglobin levels and reduce the effects of chronic anemia associated with beta thalassemia pathophysiology.
However, repeated transfusions lead to iron overload, as the body has no natural way to eliminate excess iron. To prevent iron accumulation in vital organs such as the heart and liver, patients must follow strict iron chelation therapy. Chelation drugs bind excess iron and facilitate its removal through urine or feces, significantly reducing the risk of organ damage.
Splenectomy
In many patients, the spleen becomes enlarged due to increased destruction of abnormal red blood cells—a direct consequence of beta thalassemia pathophysiology. When this process becomes severe, a splenectomy (surgical removal of the spleen) may be recommended.
While this procedure can reduce the need for frequent transfusions, it also increases susceptibility to infections. Therefore, patients require lifelong monitoring and appropriate vaccinations to minimize risks.
Bone Marrow Transplantation and Gene Therapy
At present, the only established cure for beta thalassemia is hematopoietic stem cell transplantation (bone marrow transplant) from a compatible donor. This treatment can correct the defective blood-forming system, directly addressing the root cause of beta thalassemia pathophysiology. However, it is limited by donor availability, high cost, and potential complications.
Gene therapy represents the most promising advancement in treatment. Innovative approaches involve modifying a patient’s own stem cells by inserting a functional HBB gene or using advanced gene-editing technologies like CRISPR to repair the mutation. These corrected cells are then reintroduced into the body, offering the potential for long-term or even permanent correction of the disease.
Additional Key Point: Supportive and Preventive Care
An essential aspect of managing beta thalassemia pathophysiology includes supportive care strategies such as regular monitoring, folic acid supplementation, infection prevention, and management of complications like osteoporosis and endocrine disorders. Comprehensive care ensures better long-term outcomes and enhances patient quality of life.
Frequently Asked Questions
1. What is beta thalassemia pathophysiology?
Beta thalassemia pathophysiology explains how reduced or absent beta-globin production disrupts hemoglobin formation. This imbalance leads to anemia, ineffective erythropoiesis, and red blood cell destruction.
2. Why does beta-globin deficiency cause anemia?
Without enough beta-globin, hemoglobin cannot form properly. This reduces the oxygen-carrying capacity of red blood cells, leading to chronic anemia.
3. What happens to excess alpha-globin chains?
Excess alpha-globin chains become unstable and form toxic aggregates. These damage red blood cells and lead to their premature destruction.
4. What is ineffective erythropoiesis in beta thalassemia?
It refers to the destruction of red blood cell precursors in the bone marrow before they mature. This reduces the number of functional red blood cells in circulation.
5. How does oxidative stress affect red blood cells?
Oxidative stress damages the red blood cell membrane, making cells fragile. This leads to early breakdown and worsens anemia.
6. Why does bone marrow expand in beta thalassemia?
The body tries to compensate for anemia by producing more red blood cells. This causes bone marrow expansion, which can lead to skeletal deformities.
7. What is the role of erythropoietin in this condition?
Erythropoietin is a hormone released by the kidneys to stimulate red blood cell production. In beta thalassemia, its levels increase significantly due to chronic anemia.
8. What is extramedullary hematopoiesis?
It is the production of blood cells outside the bone marrow, mainly in the liver and spleen. This happens when the marrow cannot meet the body’s needs.
9. How does beta thalassemia affect organ function?
Chronic anemia and iron overload can damage organs like the heart, liver, and endocrine glands over time.
10. Can understanding pathophysiology improve treatment?
Yes, understanding beta thalassemia pathophysiology helps guide treatments like transfusions, iron chelation, and emerging gene therapies.